 A corollary to Carnot's theorem states that: All reversible engines operating between the same heat reservoirs are equally efficient. Thus, Equation 3 gives the maximum efficiency possible for any engine using the corresponding temperatures. Irreversible systems and losses of energy (for example, work due to friction and heat losses) prevent the ideal from taking place at every step.Ĭarnot's theorem is a formal statement of this fact: No engine operating between two heat reservoirs can be more efficient than a Carnot engine operating between those same reservoirs. For this figure, the curve indicates a vapor-liquid equilibrium ( See Rankine cycle). This change is indicated by the curve on a T– S diagram. The entropy of a real material changes with temperature. Carnot's theorem įigure 6: A real engine (left) compared to the Carnot cycle (right). The P– V diagram of the reversed Carnot cycle is the same as for the Carnot heat-engine cycle except that the directions of the processes are reversed. Heat is absorbed from the low-temperature reservoir, heat is rejected to a high-temperature reservoir, and a work input is required to accomplish all this. This time, the cycle remains exactly the same except that the directions of any heat and work interactions are reversed. That is, all the processes that compose it can be reversed, in which case it becomes the Carnot heat pump and refrigeration cycle. 
This thermal energy is the cycle initiator.Ī Carnot heat-engine cycle described is a totally reversible cycle. This is the Carnot heat engine working efficiency definition as the fraction of the work done by the system to the thermal energy received by the system from the hot reservoir per cycle. 
In a Carnot cycle, a system or engine transfers energy in the form of heat between two thermal reservoirs at temperatures T H. By Carnot's theorem, it provides an upper limit on the efficiency of any classical thermodynamic engine during the conversion of heat into work, or conversely, the efficiency of a refrigeration system in creating a temperature difference through the application of work to the system. A Carnot cycle is an ideal thermodynamic cycle proposed by French physicist Sadi Carnot in 1824 and expanded upon by others in the 1830s and 1840s. 'Revisiting The Second Law of Energy Degradation and Entropy Generation: From Sadi Carnot's Ingenious Reasoning to Holistic Generalization'. Kittel, Charles Kroemer, Herbert (1980).Halliday, David Resnick, Robert (1978). Cycle de carno exercices archive#(1910) The Steam-Engine and Other Engines edition 3, page 62, via Internet Archive Carnot, Sadi, Reflections on the Motive Power of Fire.'Cycling Tames Power Fluctuations near Optimum Efficiency'. ^ Holubec Viktor and Ryabov Artem (2018).College Physics: Reasoning and Relationships. 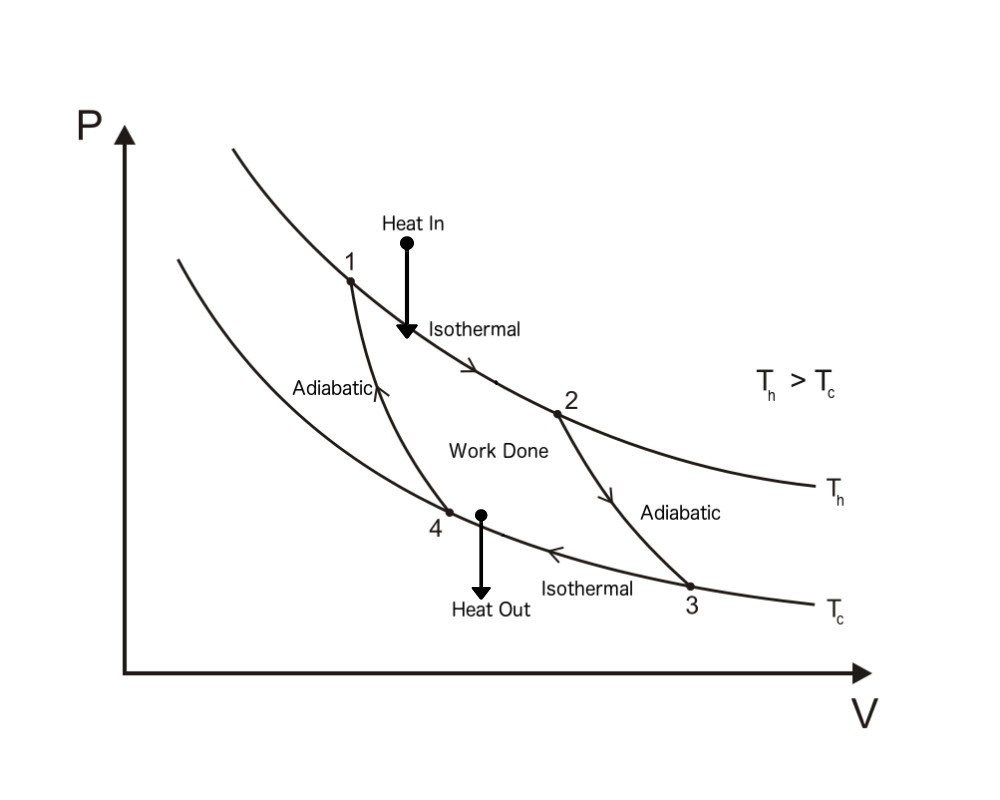
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
